|
11/23/2023 0 Comments Half life of second order reaction
Determination of bulk density, true density and porosity.Determination of particle size, and particle size distribution using the Microscopic method.Determination of particle size, and particle size distribution using the sieving method.The half -life (t1/2) of the given ester (methyl acetate or ethyl acetate) in 0.025N NAOH at room temperatureįrom graphical method= From substitution method= The reaction rate constant (K2) of the given ester (methyl acetate or ethyl acetate) in 0.025N NAOH at room temperatureįrom graphical method= minutes From substitution method= minutes Draw a plot by taking x/a (a-x) on y-axis and time on x-axis. 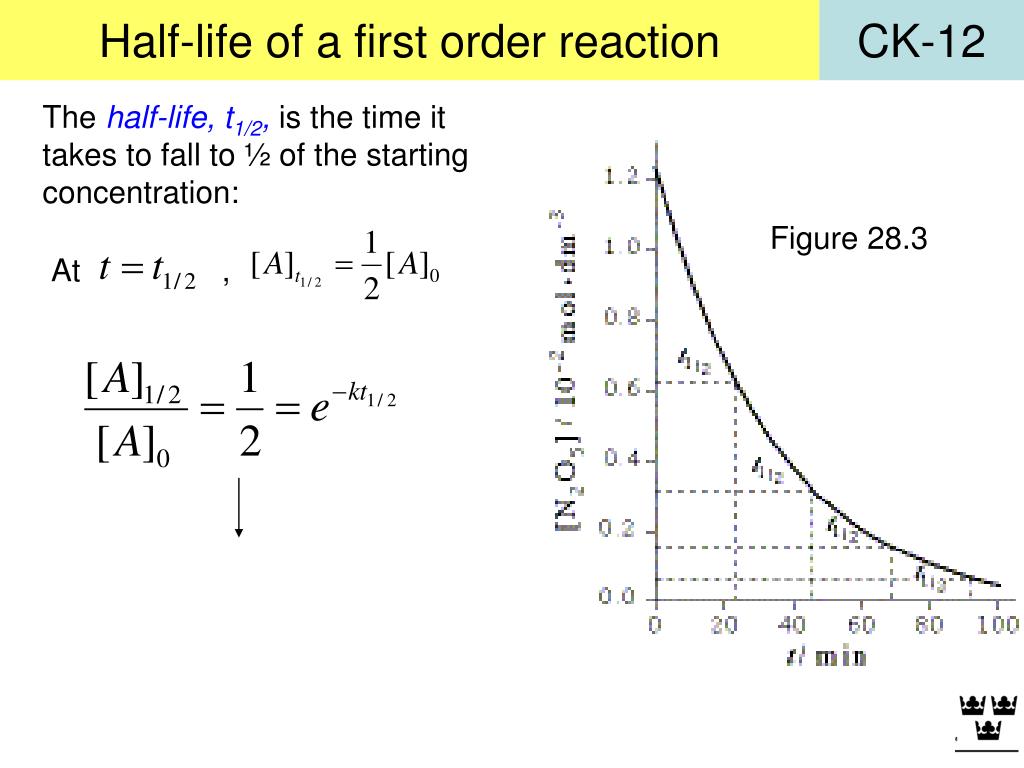
Calculate the average of the reaction rate constant (K2).Substitute the values in integral equation and calculate the reaction rate constant (K2).These titter values denote the amount of sodium hydroxide or ethyl acetate remain unreacted ie, (a-x) at time. Periodically withdrawn samples at 5, 10, 15, 20, 25, 30 minutes time periods.This titter value times t=0 corresponds to the original concentration “a” report the results Titrate against 0.02N hydrochloric acid.Add few drops of Phenolphthalein indicator.Immediately after mixing, withdrawn a 10ml sample of the mixture with pipette and transfer into a conical flask containing 10ml ice cold water.Mix the alkali and ester solution thoroughly and keep in same water bath.Keep it in above water bath for equilibrium. Measure 50ml of 0.05N of the given ester and transfer into a conical flask.Keep it in a water bath for equilibrium at room temperature. Measure 50ml of 0.05N sodium hydroxide solution and transfer into a conical flask.allow it to stand overnight and pour off the clear liquid into a bottle. Add water slowly with continuous stirring, while cooling the flask under running tap water. Weigh 2.0gm of sodium hydroxide in water and transfer into 1000ml volumetric flask. Preparation of sodium hydroxide solution (0.05N) Add 1.7ml of concentrated hydrochloric acid slowly and shake. Measure 850 ml of distilled water into 1000ml volumetric flask. Solutions of any normality XN may be prepared by diluting 85 xml of hydrochloric acid to 1000ml with water. 
Preparation of hydrochloric acid solution (0.02N) Measure 50 ml of Ethyl acetate and transfer into 1000ml volumetric flask dilute to 1000ml with distilled water. The molecularity weight of ethyl acetate is 88.10 density is 0.90g/ml.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
